 If there is an adiabatic free expansion of a gas, the temperature is unchanged (isothermal) so you might think. Relationship between temperature and Entropy: A system at a higher temperature has greater. into the gas) there is an increase in entropy of the gas. Entropy is higher as the disorders in the isolated system increase. Since entropy is dQ/T, and dQ is positive (ie. internal energy remains constant) there must be heat flow into the gas. Stars are hotter than planets, for example, which are warmer than icy asteroids, which are warmer still than the vacuum of the space between them. From the first law, if temperature remains the same (ie. The entropy of a substance changes during a change in state The entropy increases when a substance melts (change from solid to liquid) The entropy increases. If you increase temperature, more energy put into a system excites the molecules and the amount of random activity, hence the entropy. 
Although it may have seemed disorderly, and therefore, superficially entropic, in fact, there was enormous potential energy available to do work-all the future energy in the universe.Īs the universe matured, temperature differences arose, which created more opportunity for work. Gravity played a vital role in the young universe. 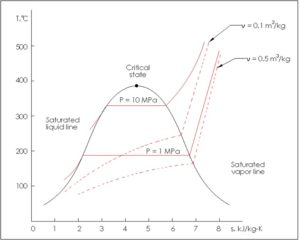
In the early, energetic universe, all matter and energy were easily interchangeable and identical in nature. I know that the entropy of a substance increases at higher temperatures but I do not know how this correlates to the situation of a reaction since I'd assume that both the reactants and the products would be at a higher entropy. Heat Death of the Universe: An Overdose of Entropy 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
